Akelos Inc.
An Emerging Leader in Treating Neuropathic Pain
An innovative research-based biotechnology company
Akelos Inc. is a biopharmaceutical clinical company focused on the translation of innovative science into treatment. The company currently is developing novel non-narcotic drugs for the treatment of neuropathic pain. The goal of Akelos is to address some of today’s most pressing areas of unmet needs.
Download the Akelos Presentation
Building New Frontiers to Address Neuropathic Pain
Akelos is an innovative research-based biotechnology company founded to address the Opioid epidemic through opioid alternatives, with drug candidates in pre-clinical development. Akelos has proudly entered into a research collaboration to develop and commercialize a novel, non-opioid anti-hyperalgesic drug to treat chronic and neuropathic pain.
Treatment guidelines for neuropathic pain include gabapentin or pregabalin, antidepressants, lidocaine patches, and tramadol. Less than 35% of patients achieve meaningful benefits from current therapeutic approaches. 3rd line treatments are dangerous opioids. While these drugs are less-frequently prescribed in recent years, are still a major threat, in particular to patients who choose to self-medicate in order to alleviate their pain. We believe it's time to pursue safe and effective treatments.
Meet Our Team

Akelos is a collaboration founded to develop opioid drug alternatives.
Researchers at Weill Cornell University have developed novel compounds for the treatment of neuropathic pain, resulting in intellectual property that is owned by Cornell. Akelos has licensed this intellectual property and is developing a treatment for neuropathic pain using the novel compounds.
"The unprecedented opioid epidemic has made it all the more important to develop new medications that relieve pain without the addictive qualities that can lead to dependency. Our medical research, led by Dr. Goldstein, is a critical step to realizing the goal of an opioid alternative." - Dr. Steven Fox, Chairman of Akelos Inc.
Published Data
Akelos Publication in the British Journal of Anaesthesia
A non-opioid designer molecule for treating chronic neuropathic pain by calming hyperactive pain-sensing neurons in the peripheral nervous system has had promising results in a preclinical study conducted by researchers at Weill Cornell Medicine and the Burke Neurological Institute...
View MoreAkelos Publication in Nature
Hyperpolarization-activated cyclic nucleotide-gated (HCN) channels are essential for pacemaking activity and neural signalling . Drugs inhibiting HCN1 are promising...
View MoreResearch Findings
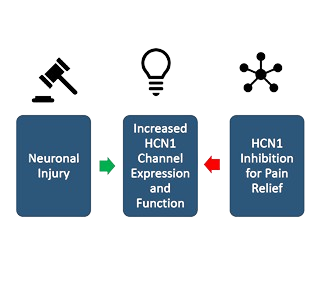
Primary sensory neurons play a pivotal role in modulating neuropathic pain
When sensory neurons are injured, HCN1 activity is enhanced
Akelos’ compounds are intended to selectively and potently inhibit HCN1 channel activity to limit the experience of pain
Akelos in the News
Akelos Inc. Receives Chinese Patent For Its Lead Compound
Akelos Inc. . . Announces that the China National Intellectual Property Administration (China’s patent office) has allowed a patent
View MoreAkelos Inc. Announces Japan Patent Office Allowance Of Patent Claiming its Lead Compound
Akelos Inc., a biotechnology company currently developing and commercializing a novel, non-opioid anti- hyperalgesic drug...
View MoreAlkylphenol Binding Site Identified in Human HCN1 Channels
In a just published study in the journal Nature, three research teams identify the binding site in human HCN1 (hHCN1) ion channel...
View MoreScott L. Dax, MS, PhD, Research and Development Champion, Joins Scientific Advisory Board of Akelos Inc.
Akelos Inc. Appoints Dr. Scott L. Dax to Scientific Advisory Board, Furthering Research and Development of Non-Opioid Solutions for Chronic...
View MoreThe Exit: Raising $70 Million and Going Public with Dr. Steve Fox
This is an incredible story of Dr. Steve Fox bringing together some of the best minds in the country to change the world of teeth hygiene forever...
View MoreCyrus Arman, PhD MBA, Joins Akelos Inc., as Head of Business Development
Akelos Inc., a biotechnology company currently developing and commercializing a novel, non-opioid anti-hyperalgesic drug to treat...
View MoreTransforming Dentistry and Beyond: A Conversation with Dr. Steven Fox
In this episode, we delve into Dr. Fox’s groundbreaking work in dentistry, where he transformed the industry with innovative oral...
View MoreTransforming Dentistry and Beyond: A Conversation with Dr. Steven Fox
In this episode, we delve into Dr. Fox’s groundbreaking work in dentistry, where he transformed the industry with innovative oral...
View MoreAkelos Study in The British Journal of Anesthesia
Potential Neuropathic Pain Treatment Shows Promise in Preclinical Tests. A non-opioid designer molecule for treating chronic neuropathic ...
Akelos Inc. Announces United States Patent and Trademark Office Allowance of Patent Claiming its Lead Compound
NEW YORK, NEW YORK, UNITED STATES, February 14, 2023 /EINPresswire.com/ -- Akelos Inc., announces that the United States Patent and Trademark Office has allowed a patent application (17/254,787). . .
View MoreDarryle D. Schoepp, Ph.D., Former Merck and Eli Lilly Executive, Joins Scientific Advisory Board of Akelos Inc., Biotechnology Company
NEW YORK, February 1, 2023 (Newswire.com) - Akelos Inc., a biotechnology company currently developing and commercializing a novel, non-opioid anti-hyperalgesic drug to treat ...
Akelos Inc. Receives Prestigious Grant From NINDS of the National Institute of Health
Akelos Inc. has been awarded a Phase I/II Fast-Track Small Business Technology Transfer (STTR) grant. . .
Akelos Inc. is named one of the 29 Best New York City Biopharma Companies and Startups
Akelos is a new biopharmaceutical company focused on the translation of innovative science into treatment.
Akelos Inc.'s SAB Member Co-Authors Paper Validating HCN1 as a Therapeutic Target for the Treatment of Peripheral Neuropathic Pain
NEW YORK, February 5, 2021 (Newswire.com) - Dr. Peter Goldstein, scientific co-founder . . .
Akelos Inc.'s Lead Compound Achieves Preclinical Milestone
. . .These results clearly validate the lead molecule as an effective approach to relieving neuropathic pain, which is notoriously difficult to treat. That it is a non-opiate only enhances its usefulness. . .
Akelos Inc.'s PCT International Patent Application, Licensed From Cornell University, Has Published
Akelos Inc., a biotechnology company currently developing and commercializing a novel, non-opioid anti-hyperalgesic drug to treat chronic and neuropathic . . .
Thomas G. Heffner, Ph.D., Former Pfizer Executive, Joins Akelos Inc. as a Strategic Advisor
NEW YORK, November 18, 2019 (Newswire.com) - Akelos Inc., a biotechnology company currently developing and commercializing a novel, non-opioid . . .
Roger Frechette, Ph.D., Joins Akelos Inc., as Director of Operations & Business Development
NEW YORK, June 17, 2019 (Newswire.com) - Akelos Inc., a biotechnology company currently developing and commercializing a novel, non-opioid . . .
Charles B. Berde, MD, PhD, Joins Scientific Advisory Board of Akelos Inc., Biotechnology Company Developing Opioid Alternatives
NEW YORK, April 29, 2019 (Newswire.com) - Akelos Inc., a biotechnology company currently developing and commercializing ...
J. David Warren, Ph.D., Joins Scientific Advisory Board of Akelos Inc., Biotechnology Company Developing Opioid Alternatives
NEW YORK, April 25, 2019 (Newswire.com) - Akelos Inc., a biotechnology company currently developing and commercializing a ...
Akelos Inc. Identifies Key Aspects Governing Pharmacophore Interactions With Target Site
NEW YORK, February 25, 2019 (Newswire.com) - Akelos Inc., a biotechnology company currently developing and commercializing a novel, non-opioid anti-hyperalgesic drug to treat chronic ...
Akelos Inc. Files Third Patent Application to Develop New Treatment Option for Neuropathic Pain
NEW YORK, February 11, 2019 (Newswire.com) - Akelos Inc., a biotechnology company currently developing and commercializing a novel, non-opioid anti-hyperalgesic drug to treat ...
Get In Touch
Contact Us
Location
215 East 68th Street, Suite 26-B
New York, NY 10065
Phone Number
(212) 953 - 1544Social Media
















